 In this new electron configuration, the highest occupied energy level is no longer indicated by a 4. The following electron configuration results: If both of the valence electrons in the 4 s orbital are lost, as shown below, Therefore, calcium will lose both of its valence electrons.Ĭonsider the impact that this will have on calcium's electron configuration. Because calcium only has two valence electrons, losing both of them is possible, as doing so would not exceed the maximum loss-limit of three electrons. Unfortunately, the valence electron count of all metals renders them unable to gain enough electrons to achieve an octet configuration.Īlternatively, an atom can lose its valence electrons, in order to achieve an octet configuration. However, as stated above, gaining more than three electrons is energetically-unfavorable and will not occur. Therefore, calcium would need to gain 6 electrons in order to achieve an octet configuration. In order to be stable, a particle must possess an octet, or eight, fully-paired valence electrons. Therefore, the neutron count for this particular calcium atom is irrelevant to the analysis that will be performed in the following paragraphs and will not be discussed further.Ĭalcium's electron configuration, which was first determined in Exercise 2.6.1, is shown below.Ĭalcium has 2 valence electrons, as determined either by totaling the electrons found within the orbitals in the highest occupied energy level, which, in this case, is the 4 s orbital, or by identifying the "A/B System" group number for the column in which the element is found. The overall charge of a particle is not influenced by the number of neutrons that are present, as neutrons are uncharged, by definition. Recall that the number of neutrons present in an atom can vary, depending on which isotope of calcium is being considered. The resultant particle is instead is classified as an ion, as it has become a charged particle.Īs stated previously, cations are positively-charged ions that are most often formed when metals, which are found on the left side of the periodic table, lose valence electrons.Īn atom of calcium contains 20 protons, because its atomic number is 20, and 20 electrons, in order to be net-neutral. Thus, the term "atom" no longer applies to this new particle, since an atom must be net-neutral, by definition. As a result, this new particle will no longer be neutral, but rather will bear a net charge. As a result, inner shell electrons do not participate in the ionization process.īy changing the number of electrons that are contained within a particle, an imbalance is created between the number of positively-charged protons and negatively-charged electrons that it possesses. Finally, note that inner shell electrons are always fully-paired and, therefore, are considered stable. Gaining or losing more than this number of electrons is energetically-unfavorable and will not occur. The remaining elements must, therefore, gain or lose up to three valence electrons, in order to achieve an octet configuration. 
Only the noble gases naturally have an octet configuration, meaning that they possess eight, fully-paired valence electrons. 
Determine the charges achieved when main group metals ionize.Īs stated previously, electrons are most stable when they exist in pairs.Explain how neutral atoms ionize to form cations.A fun video showing potassium metal and its reactivity can be found HERE.\) It is used to create this same effect and color in fireworks. Potassium burns with a purple flame and throws purple sparks. The heat of this reaction ignites the hydrogen gas that is also produced from the water. Pure potassium metal reacts explosively when placed in water as it donates its valence electron to oxygen. (Only the 3 outer shells are depicted here.) Neutral potassium (K) atom (L) compared to the much smaller potassium (K +) ion (R). That is why potassium forms a 1+ ionic state so violently. With a lower ionization energy, potassium will give up its valence electron more eagerly than sodium in an ionic interaction, in order to reach the stability of the 3 s 23 p 6 noble gas configuration of argon, which is a multi-di-electron state with three concentric full shells. These orbitals represent phase-locked, resonant, coherent, harmonic, stationary waves. CLICK HERE to interact with this object.Īs we saw in the case of argon, the 3 rd shell orbitals are more like spherical tetrahedra, and the 4 th shell is a single electron in a spherical s-orbital. 
Being one shell larger than sodium, potassium has a lower ionization energy and is therefore more reactive. Potassium has the same electron configuration as sodium, but with three full shells within that have the identical configuration to argon. It has 19 protons and 20 neutrons in the nucleus, giving it a mass of 39 amu, and it has 19 electrons enveloping the nucleus. 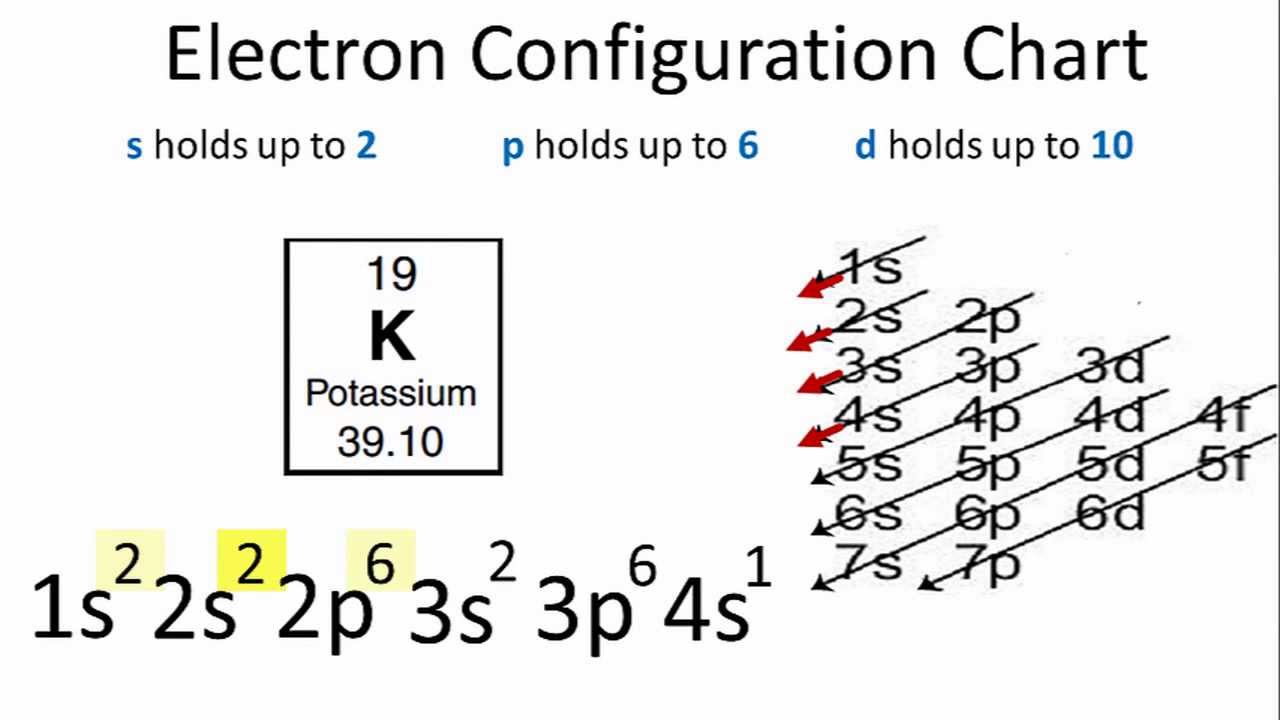
Potassium is the 19 th element on the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
